The Clickable
Guard Cell: Electronically linked
Model of Guard Cell Signal Transduction Pathways
Pascal Mäsera, Nathalie Leonhardtb,c and Julian I. Schroederb1
aInstitute of Cell Biology, University of Berne, CH-3012 Bern, Switzerland
bDivision of Biological Sciences, Cell and Developmental Biology Section and Center for Molecular Genetics,
University of California, San Diego, La Jolla CA92093-0116, USA
cDepartement d'ecophysiologie vegetale et de microbologie, CEA Cadarache, F-13180 St Paul Lez
Durance, France
1Corresponding author, e-mail: juilan at biomail dot ucsd dot edu, phone: 858-534-7759, fax: 858-534-7108
The Arabidopsis Book, eds. C. R. Somerville
& E. Meyerowitz, ASPB
Abstract
Guard cells are located in the leaf epidermis and pairwise surround
stomatal pores, which allow CO2 influx for photosynthetic carbon fixation
and water loss via transpiration to the atmosphere. Signal transduction
mechanisms in guard cells integrate a multitude of different stimuli
to modulate stomatal aperture. Stomata open in response to light. In
response to drought stress, plants synthesize the hormone abscisic acid
(ABA) which triggers closing of stomatal pores. Guard cells have become
a well-developed system for dissecting early signal transduction mechanisms
in plants and for elucidating how individual signaling mechanisms can
interact within a network in a single cell. Previous
reviews have described pharmacological modulators that affect guard
cell signal transduction. Here we focus on mechanisms for which genes
and mutations have been characterized, including signaling components
for which there is substantial biochemical evidence such as ion channels
which represent targets of early signal transduction mechanisms. Guard
cell signaling pathways are illustrated as graphically linked models:
• Figure 1 is linked to explanations that appear upon mouse-over.
• Figures 2 and 3 are clickable and linked to explanatory text for each gene and the respective signaling component. The text is an updated version of Schroeder et al. (2001), used with permission of Annual Reviews of Plant Biology.
• Genes that are discussed within this review are directly linked to electronic databases: Arabidopsis genes are linked to MAtDB, transporter families are linked to PlantsT, genes from other plant species to GenBank. Recent publications are linked to abstracts in PubMed; for older publications a list of all references is included at the end of the text.
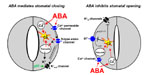
A model for roles of ion channels in ABA signalling.
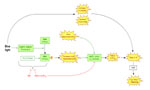
Light-induced stomatal opening.
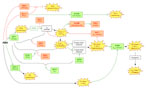
ABA-induced stomatal closing.
Introduction
Being rooted in one place plants must respond to changes in environmental conditions and stresses. Hormone and light signal transduction pathways form complex networks that control plant responses to the environment. In spite of their importance for plant growth and development, these signaling pathways remain "grey boxes", with many major players and their interactions remaining unknown. In guard cells, a network of signal transduction mechanisms integrates water status, hormone responses, light, CO2 and other environmental conditions to regulate stomatal movements in leaves for optimization of plant growth and survival under diverse conditions. Stomatal guard cells have become one of the well-developed model systems for understanding how various signaling components can interact within a network in a single plant cell. Guard cells allow quantitative dissection of the functions of individual genes and proteins within signaling cascades because:
1. Guard cells respond cell-autonomously to physiological stimulation, allowing cell biological analyses of stomatal opening and closing in response to various stimuli.
2. Guard cells respond to most of the classically known hormonal stimuli in plants, illustrating that presently unidentified but essential plant receptors and early signalling mechanisms function in these cells.
3. A network of ion channels in the plasma membane and tonoplast of guard cells has been characterized that, together with metabolic responses, controls stomatal movements (see Figure 1). Being targets of early signaling branches these ion channels provide effective probes to identify and characterize upstream regulators.
4. Stomatal aperture regulates plant CO2 intake and water loss, thus critically influencing growth and water stress responsiveness. In addition, powerful techniques have recently been developed and adapted for guard cell signal transduction studies, allowing interdisciplinary, molecular genetic, cell biological, second messenger, biophysical, physiological and genomic analyses.
The roles of ion channels in ABA signaling
The hormone ABA triggers a signalling cascade in guard cells that results in stomatal closure and inhibits stomatal opening. Stomatal closure is mediated by turgor reduction in guard cells, which is caused by efflux of K+ and anions from guard cells, sucrose removal, and a parallel conversion of the organic acid malate to osmotically inactive starch (MacRobbie, 1998). Figure 1 shows an extension of early models for the roles of ion channels in ABA-induced stomatal closing (Schroeder and Hedrich, 1989; McAinsh et al., 1990).
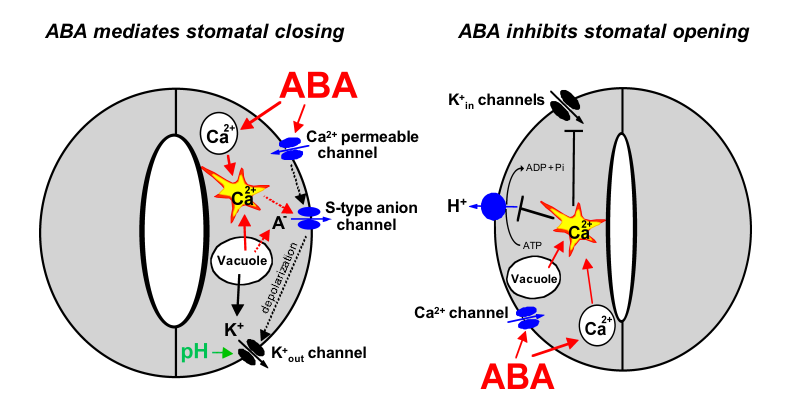
Mouse-over for explanations or see text below (mouse-over does not work on Mac browsers and some PC browsers) .
As vacuoles can take up over 90% of the guard cell’s volume, over 90% of the ions exported from the cell during stomatal closing must first be transported from vacuoles into the cytosol (MacRobbie, 1998; MacRobbie, 1995). [Ca2+]cyt elevation activates vacuolar K+ (VK) channels proposed to provide a pathway for Ca2+-induced K+ release from the vacuole (Ward and Schroeder, 1994). At resting [Ca2+]cyt, K+ efflux from guard cell vacuoles can be mediated by fast vacuolar (FV) channels, allowing for versatile vacuolar K+ efflux pathways (Allen and Sanders, 1996). The pathways for anion release from vacuoles remain elusive.
Stomatal opening is driven by plasma membrane proton-extruding H+-ATPases. H+-ATPases can drive K+ uptake via K+in channels (Fig. 1, right panel; Kwak et al., 2001). Cytosolic Ca2+ elevations in guard cells down-regulate both K+in channels (Schroeder and Hagiwara, 1989) and plasma membrane H+-ATPases (Kinoshita et al., 1995), providing a mechanistic basis for ABA and Ca2+ inhibition of K+ uptake during stomatal opening (Fig. 1, right panel).
Guard cells respond to a multitude of signals including temperature, partial CO2 pressure, light, humidity, and hormonal stimuli. For the majority of signals the molecular identity of the sensors is not known, with the notable exception of blue light: The phototropins PHOT1 and PHOT2 were shown to be the blue light receptors in Arabidopsis guard cells (Kinoshita et al., 2001). Figure 2 illustrates the signaling cascade from activation of PHOT1,2 to stomatal opening in terms of known genes and their relative position in the signal transduction pathway.
One of the best understood plant signaling networks is the one triggered by ABA in guard cells, causing stomatal closure. As shown in Figure 3, many genes encoding for positive as well as negative regulators of guard cell ABA signaling have been identified in Arabidopsis. The gene(s) encoding the ABA receptor(s), however, remained elusive. Most of the ABA signal transduction components were found by classic "forward" genetic screens for either increased or reduced sensitivity to ABA.
We thank Annual Reviews for permission to use the review by Schroeder et al. (2001) for this electronic model. We thank Dr. June Kwak for help in preparing figure 1 and Guillaume Leonhardt for help with figures 2 and 3. Preparation of this article and research from the authors' laboratory was supported by NSF (MCB 0077791) and NIH (R01GM060396) grants to J.I.S., Human Frontiers Science Program fellowships to N.L. and P.M., and in part by a grant from DOE (DE-FG02-03ER15449) and NSF (DBI 0077378) to J.I.S.
Allen, G.J., and Sanders, D. (1996) Control of ionic currents guard cell vacuoles by cytosolic and luminal calcium. Plant J. 10, 1055-1069.
Blatt, M.R., and Armstrong, F. (1993) K+ channels of stomatal guard cells: Abscisic-acid-evoked control of the outward-rectifier mediated by cytoplasmic pH. Planta 191, 330-341.
Hedrich, R., Busch, H., and Raschke, K. (1990) Ca2+ and nucleotide dependent regulation of voltage dependent anion channels in the plasma membrane of guard cells. EMBO J. 9, 3889-3892.
Kinoshita, T., Nishimura, M., and Shimazaki, K.I. (1995) Cytosolic concentration of Ca2+ regulates the plasma membrane H+-ATPase in guard cells of fava bean. Plant Cell 7, 1333-1342.
Kinoshita, T., Doi, M., Suetsugu, N., Kagawa, T., Wada, M., and Shimazaki, K. (2001) Phot1 and phot2 mediate blue light regulation of stomatal opening. Nature 414, 656-60.
Kwak, J.M., Murata, Y., Baizabal-Aguirre, V.M., Merrill, J., Wang, M., Kemper, A., Hawke, S.D., Tallman, G., and Schroeder, J.I. (2001) Dominant negative guard cell K+ channel mutants reduce inward-rectifying K+ currents and light-induced stomatal opening in Arabidopsis. Plant Physiol. 127, 473-485.
Ilan, N., Schwartz, A., and Moran, N. (1994) External pH effects on the depolarization-activated K channels in guard cell protoplast of Vicia faba. J. Gen. Physiol. 103, 807-831.
MacRobbie, E.A.C (1995) ABA-induced ion efflux in stomatal guard cells: multiple actions of ABA inside and outside the cell. Plant J. 7, 565-576.
MacRobbie, E.A.C. (1998) Signal transduction and ion channels in guard cells. Phil. Trans. Roy. Soc. London 1374, 1475-1488.
McAinsh, M.R., Brownlee, C., and Hetherington, A.M. (1990) Abscisic acid-induced elevation of guard cell cytosolic Ca2+ precedes stomatal closure. Nature 343, 186-188.
Miedema, H., and Assmann, S.M. (1996) A membrane-delimited effect of internal pH on the K+ outward rectifier of Vicia faba guard cells. J. Mem. Biol. 154, 227-237.
Schroeder, J.I., Raschke, K., and Neher, E. (1987) Voltage dependence of K+ channels in guard cell protoplasts. Proc. Natl. Acad. Sci. USA 84, 4108-4112.
Schroeder, J.I., and Hedrich, R. (1989) Involvement of ion channels and active transport in osmoregulation and signaling of higher plant cells. Trends Biochem. Sci. 14, 187-192.
Schroeder, J.I., and Hagiwara, S. (1989) Cytosolic calcium regulates ion channels in the plasma membrane of Vicia faba guard cells. Nature 338, 427-430
Schroeder, J.I., Allen, G.J., Hugouvieux, V., Kwak, J.M., and Waner, D. (2001) Guard cell signal transduction. Annu. Rev. Plant Physiol. Plant Mol. Biol. 52, 627-658
Schulz-Lessdorf, B., Lohse, G., and Hedrich, R. (1996) GCAC1 recognizes the pH gradient across the plasma membrane: A pH-sensitive and ATP-dependent anion channel links guard cell membrane potential to acid and energy metabolism. Plant J. 10, 993-1004.
Ward, J.M., and Schroeder, J.I. (1994) Calcium-activated K+ channels and calcium-induced calcium release by slow vacuolar ion channels in guard cell vacuoles implicated in the control of stomatal closure. Plant Cell 6, 669-683.